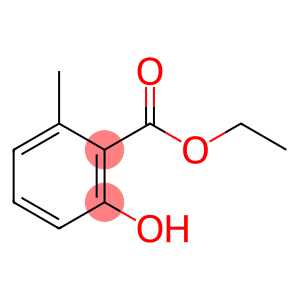
ethyl 2-hydroxy-6-methylbenzoate
Methylsalicylicacidethylester
CAS: 6555-40-4
Molecular Formula: C10H12O3
ethyl 2-hydroxy-6-methylbenzoate - Names and Identifiers
| Name | Methylsalicylicacidethylester |
| Synonyms | ETHYL 6-METHYLSALICYLATE Ethyl 6-methylsalicylate Methylsalicylicacidethylester 6-Methylsalicylsureethylester ETHYL 2-HYDROXY-6-METHYLBENZOATE ethyl 2-hydroxy-6-methylbenzoate 6-Methylsalicylic acid ethyl ester 6-METHYLSALICYLIC ACID ETHYL ESTER 2-HYDROXY-6-METHYLBENZOIC ACID ETHYL ESTER |
| CAS | 6555-40-4 |
| EINECS | 613-820-4 |
| InChI | InChI=1/C10H12O3/c1-3-13-10(12)9-7(2)5-4-6-8(9)11/h4-6,11H,3H2,1-2H3 |
ethyl 2-hydroxy-6-methylbenzoate - Physico-chemical Properties
| Molecular Formula | C10H12O3 |
| Molar Mass | 180.2 |
| Density | 1.136±0.06 g/cm3(Predicted) |
| Melting Point | 45°C |
| Boling Point | 85-95 °C(Press: 0.5 Torr) |
| Flash Point | 98.203°C |
| Vapor Presure | 0.015mmHg at 25°C |
| Appearance | Solid |
| Color | White to Light yellow |
| pKa | 10.04±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.535 |
| MDL | MFCD01194299 |
ethyl 2-hydroxy-6-methylbenzoate - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| HS Code | 29182300 |
ethyl 2-hydroxy-6-methylbenzoate - Introduction
Methylsalicylicacidethylester, the chemical formula is C9H10O3, is an organic compound. The following is a description of its nature, use, preparation and safety information:
Nature:
Methylsalicylicacidethylester is a colorless liquid with the characteristic odor of salicylic acid. It has a high density, a boiling point of 222-223°C, and a melting point of -5°C. It is soluble in many organic solvents such as ethanol, ether and benzene, and slightly soluble in water.
Use:
Methylsalicylicacidethylester has certain pharmacological activities and can be used as an over-the-counter drug. It is often used to relieve mild skin inflammation, pain and fever. It is also commonly used in the preparation of acid dyes, spices and health products.
Preparation Method:
Methylsalicylicacidethylester can be prepared by the reaction of phenol and acetic anhydride under base catalysis. First, phenol reacts with acetic anhydride to form phenyl acetate, which is then esterified with an acid catalyst in the presence of a base to convert the phenyl acetate to Methylsalicylicacidethylester.
Safety Information:
Methylsalicylicacidethylester is a relatively safe compound, but still need to pay attention to some safety measures. Avoid prolonged contact with the skin and inhalation of its vapors, such as contact with the skin should be washed immediately with water. Avoid contact with eyes and mouth during use. In addition, it should be stored in a closed container, away from fire and oxidizing agents. Proper laboratory safety procedures shall be followed during use and storage. If taken by mistake or inhaled by mistake, seek medical attention immediately.
Nature:
Methylsalicylicacidethylester is a colorless liquid with the characteristic odor of salicylic acid. It has a high density, a boiling point of 222-223°C, and a melting point of -5°C. It is soluble in many organic solvents such as ethanol, ether and benzene, and slightly soluble in water.
Use:
Methylsalicylicacidethylester has certain pharmacological activities and can be used as an over-the-counter drug. It is often used to relieve mild skin inflammation, pain and fever. It is also commonly used in the preparation of acid dyes, spices and health products.
Preparation Method:
Methylsalicylicacidethylester can be prepared by the reaction of phenol and acetic anhydride under base catalysis. First, phenol reacts with acetic anhydride to form phenyl acetate, which is then esterified with an acid catalyst in the presence of a base to convert the phenyl acetate to Methylsalicylicacidethylester.
Safety Information:
Methylsalicylicacidethylester is a relatively safe compound, but still need to pay attention to some safety measures. Avoid prolonged contact with the skin and inhalation of its vapors, such as contact with the skin should be washed immediately with water. Avoid contact with eyes and mouth during use. In addition, it should be stored in a closed container, away from fire and oxidizing agents. Proper laboratory safety procedures shall be followed during use and storage. If taken by mistake or inhaled by mistake, seek medical attention immediately.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 6-METHYLSALICYLIC ACID ETHYL ESTER Request for quotation
CAS: 6555-40-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6555-40-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Ethyl 2-hydroxy-6-methylbenzoate Visit Supplier Webpage Request for quotationCAS: 6555-40-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxy-6-Methylbenzoic Acid Ethyl Ester Visit Supplier Webpage Request for quotation
CAS: 6555-40-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6555-40-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 6-METHYLSALICYLIC ACID ETHYL ESTER Request for quotation
CAS: 6555-40-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6555-40-4
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Ethyl 2-hydroxy-6-methylbenzoate Visit Supplier Webpage Request for quotationCAS: 6555-40-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxy-6-Methylbenzoic Acid Ethyl Ester Visit Supplier Webpage Request for quotation
CAS: 6555-40-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6555-40-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


